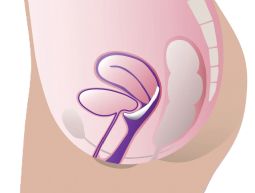
ProFem Marland Pessary
With or without a support membrane, it is used to treat symptoms of second- or third-degree uterine prolapse and cystocele. The raised ring may also help relieve stress incontinence. Holes in the membrane ease removal by releasing vacuum. Starting with a fitting set is recommended.
Classification
06 04 03 01 - Abdominal muscle supports
Documents
The product series contains 14 products.
Product 1 of 14
ProFem Marland size 2, 58 mm
HMI-no.
145790
Article-no.
M2.25-2
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145790
EU declaration of conformity for HMI-no. 145790Product 2 of 14
ProFem Marland size 3, 64 mm
HMI-no.
145793
Article-no.
M2.50-3
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145793
EU declaration of conformity for HMI-no. 145793Product 3 of 14
ProFem Marland size 4, 70 mm
HMI-no.
145796
Article-no.
M2.75-4
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145796
EU declaration of conformity for HMI-no. 145796Product 4 of 14
ProFem Marland size 5, 76 mm
HMI-no.
145799
Article-no.
M3.00-5
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145799
EU declaration of conformity for HMI-no. 145799Product 5 of 14
ProFem Marland size 6, 83 mm
HMI-no.
145802
Article-no.
M3.25-6
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145802
EU declaration of conformity for HMI-no. 145802Product 6 of 14
ProFem Marland size 7, 89 mm
HMI-no.
145805
Article-no.
M3.50-7
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145805
EU declaration of conformity for HMI-no. 145805Product 7 of 14
ProFem Marland size 8, 95 mm
HMI-no.
145808
Article-no.
M3.75-8
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145808
EU declaration of conformity for HMI-no. 145808Product 8 of 14
ProFem Marland with support size 2, 58 mm
HMI-no.
145811
Article-no.
M2.25S-2
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145811
EU declaration of conformity for HMI-no. 145811Product 9 of 14
ProFem Marland with support size 3, 64 mm
HMI-no.
145814
Article-no.
M2.50S-3
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145814
EU declaration of conformity for HMI-no. 145814Product 10 of 14
ProFem Marland with support size 4, 70 mm
HMI-no.
145817
Article-no.
M2.75S-4
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145817
EU declaration of conformity for HMI-no. 145817Product 11 of 14
ProFem Marland with support size 5, 76 mm
HMI-no.
145820
Article-no.
M3.00S-5
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145820
EU declaration of conformity for HMI-no. 145820Product 12 of 14
ProFem Marland with support size 6, 83 mm
HMI-no.
145823
Article-no.
M3.25S-6
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145823
EU declaration of conformity for HMI-no. 145823Product 13 of 14
ProFem Marland with support size 7, 89 mm
HMI-no.
145826
Article-no.
M3.50S-7
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145826
EU declaration of conformity for HMI-no. 145826Product 14 of 14
ProFem Marland with support size 8, 95 mm
HMI-no.
145829
Article-no.
M3.75S-8
Registration date
05-09-2025
Last updated
29-01-2026
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
 EU declaration of conformity for HMI-no. 145829
EU declaration of conformity for HMI-no. 145829